Current projects in the ESRG
Biophysics of metal ion coordination
Metal ion coordination in biologal systems has been an area of interest for the ESRG for the past 15 years. We have studied a wide-range of systems using calorimetry and other biophysical methods.
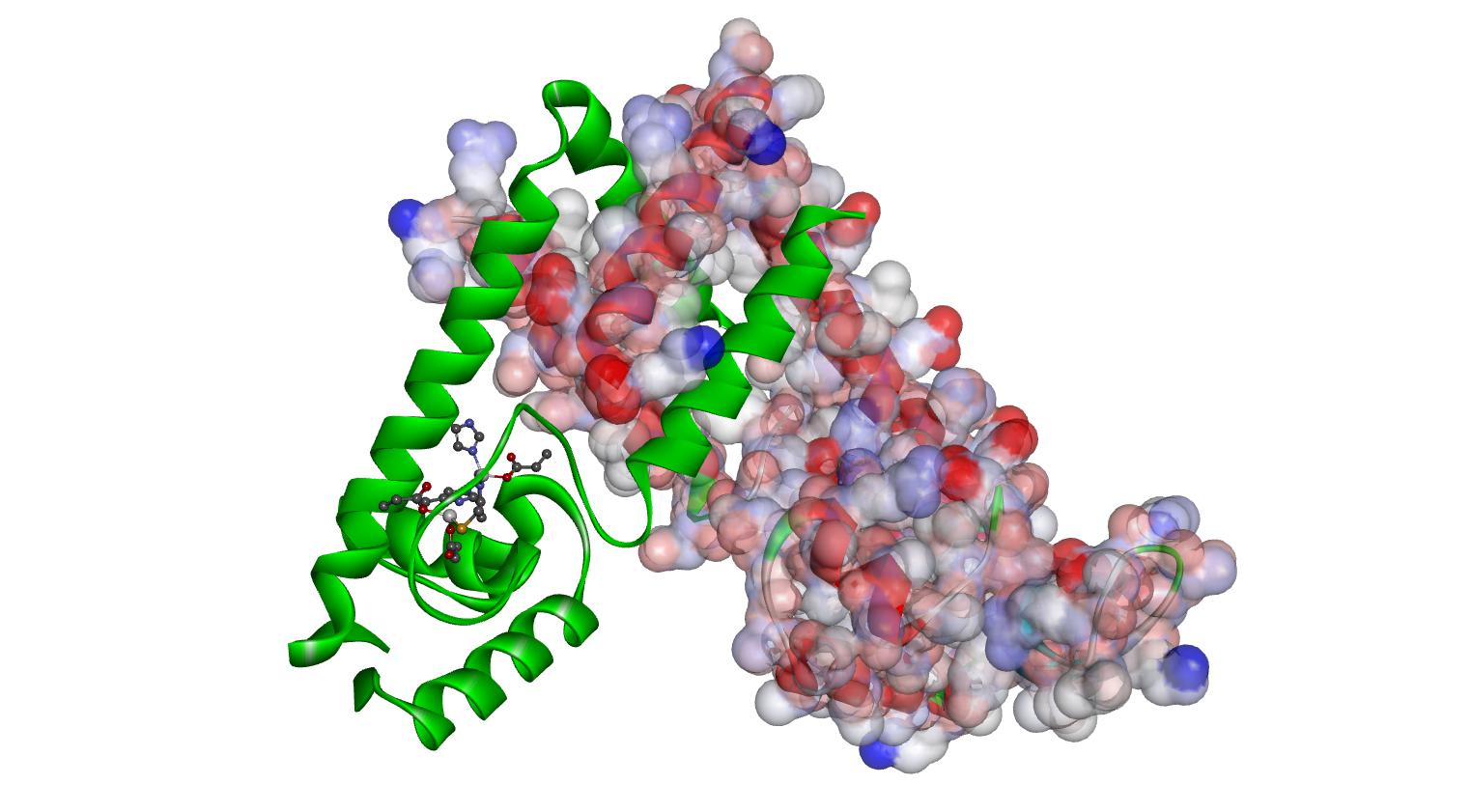
At first glance metal ion coordination in biological systems seems to be a fairly simple idea. However, these complex ion equilibria are not simple in vitro and are amazingly complex in vivo. We are actively developing new methods to explore these processes more accurately, where we aim to understand how binding of metal ions to biomolecules or small molecules to metalloproteins impacts local dynamics. Traditionally we have focused on model systems like human carbonic anhydrase II and a range of non-heme iron proteins. However, recently the ESRG has focused in on three metal-dependent transciption factors from S. pneumoniae.
At first glance metal ion coordination in biological systems seems to be a fairly simple idea. However, these complex ion equilibria are not simple in vitro and are amazingly complex in vivo. We are actively developing new methods to explore these processes more accurately, where we aim to understand how binding of metal ions to biomolecules or small molecules to metalloproteins impacts local dynamics. Traditionally we have focused on model systems like human carbonic anhydrase II and a range of non-heme iron proteins. However, recently the ESRG has focused in on three metal-dependent transciption factors from S. pneumoniae.
Recent highlights
06/01/2024

NIH CoBRE program at MSU has invested in a MicroCal PEAQ-ITC that will is housed in the ESRG research space. This system bring new opportunity for training in metal ion binding to biomolecules and the related biophysics to the group!
01/01/2021
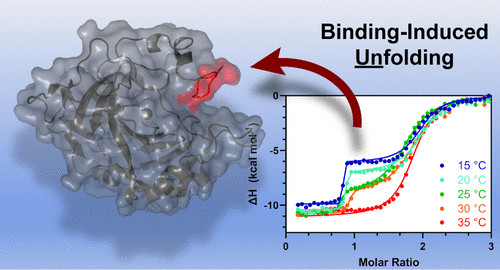
K. McConnell, N. Fitzkee, and J. Emerson have reported their efforts to understand how metal ions impact conformational dynamics in a model protein system in Inorganic Chemistry. This marks the group's first effort to look beyond metal ion binding thermodynamics and into how the protein is changing.
First row transition metal catalysts for novel reactions
In the Emerson Stokes Research Group, we are working towards designing new catalysts for sustainable novel organic transformations. This includes projects aimed at developing new ligands for organometallic complexes, utilizing first-row transition metal catalysts for C-N bond forming reactions, and the efficient, low-cost modifications of known bioactive molecules. Our ligand design has involved both N-heterocyclic carbenes (NHCs) and phenanthroline-based architectures for the arylation of small molecules and biomolecules. We are interested in exploring the mechanism and redox chemistry involved in many of these catalytic processes as well as understanding the spectroscopic changes that emerge from catalyst turnover. In addition, some of our research focus is on devising new methods of heterogeneous and homogeneous catalysis including the use of “soft materials” such as proteins, DNA, and lipids as flexible media to support novel transformations.
![crystal structure of [Cu(phen)(OTf)]+ and DFT derived HOMO](Files/Image/Other%20images/IMG_003.jpg)
Recent highlights
5/20/2025
Grad. students Teimouri and Attarroshan both reported 1st author papers in Tetrahedron Letters!
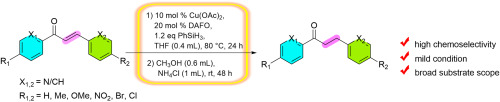
Tetrahedron Lett. 2025, 162. 155588
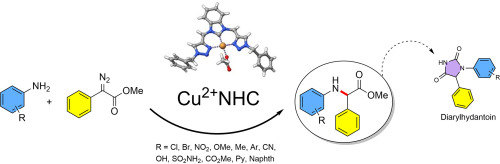
Tetrahedron Lett. 2025, 161. 155567
12/20/2024
Adhikari publishes work on monitoring the electronic impact of pyridinyl substituted NCN Cu pincer complexes.

O2 activation and interesting biological transformations
Since the start of the ESRG, we have been interested in novel reactions catalyzed by (metallo)enzymatic systems. From C-H bond activation in non-heme proteins to oxidative C-C bond cleavage Nature has interesting and unique ways of catalyzing reactions, and it is our hypothesis that these transformations can be used to catalyzed industrial processes in a sustainable fashion.
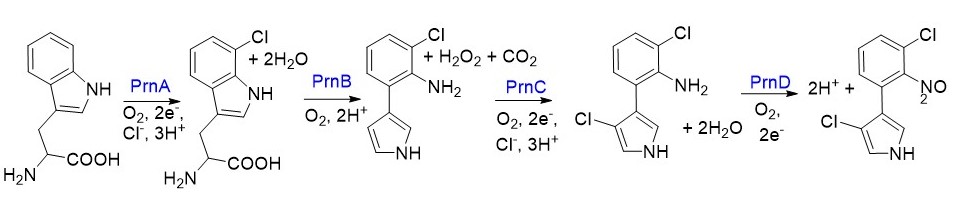
Our most recent project is focused on pyrrolnitirn biosynthesis, where there are 4 dioxygen-dependent oxidative steps that convert tryptophan into pyrrolnitirn. We are primarily interested in substrate selectivity in flavin-dependent halogenases, and how proteins like PrnA and PrnC can be adapted for commerical purposes.
Our most recent project is focused on pyrrolnitirn biosynthesis, where there are 4 dioxygen-dependent oxidative steps that convert tryptophan into pyrrolnitirn. We are primarily interested in substrate selectivity in flavin-dependent halogenases, and how proteins like PrnA and PrnC can be adapted for commerical purposes.
Recent highlights
06/01/2023
Power joins AFRL MLRCP summer internship at Wright-Patterson AFB. Her project is aimed at learning theoritical tools like molecular dynamics to preduct molecular binding modes in PrnA.


